Contact Admission
International Collaboration
Challenges in selecting antibiotics treatment for H. Pylori in Vietnam
4Pham Ngoc Thach University of Medicine, 5Phan Chau Trinh University
BACKGROUND
Antibiotics recommended in the eradication therapy for H. pylori include: Amoxicillin, Metronidazole, Clarithromycin, Levofloxacin, Tetracycline. However, in Vietnam, these antibiotics have been reported resistant by
H. pylori, particularly Metronidazole, Clarithromycin and Levofloxacin. Therefore in Vietnam, the antibiotic resistance of H. pylori is a major challenge in the use and development of therapeutic regimens for H. pylori eradication.
AIMS
Analyze the results of microbiological and molecular biology tests for H. pylori insolates in more than two years from 2015 to 2017 to make the following observations: (1) Percentage of H. pylori with virulence via the detection of cagA and vacA genes; (2) Resistance of the H. pylori isolates to 5 antibiotics including Amoxicillin, Metronidazole, Clarithromycin, Levofloxacin, Tetracycline; (3) levels of clarithromycin by analyzing the MIC results of this antibiotics on the H. pylori isolates; (4) and finally determine the CYP2C19 genotype of the patients to help doctors maximize their antibiotic treatment efficacy by selecting PPIs and appropriate PPI doses.
MATERIALS – METHODS
Samples are gastro-duodenal ulcers biopsy obtained through endoscopy. H. pylori was isolated on agar plate supplemented with 10% horse blood and with antibiotic and incubate micro-aerobic at 37 ° C for 4 days. Antibiotics susceptibility testing of Amoxicillin, Tetracycline, Metronidazole, Clarithromycin and Levofloxacin were obtained by culture of H. pylori isolates on the media plate with and without antibiotic at breakpoint concentrations. For Clarithromycin, the Etest were also used to determine MIC. Real-time PCR techniques were also performed directly on the samples to detect the cagA gene and the vacA gene of H. pylori and to identify the patient's CYP2C19 genotype.
RESULTS - DISCUSSIONS
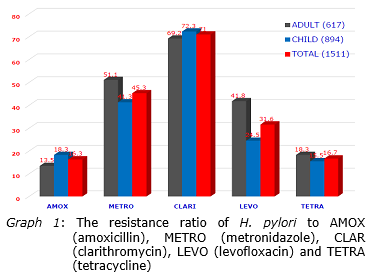
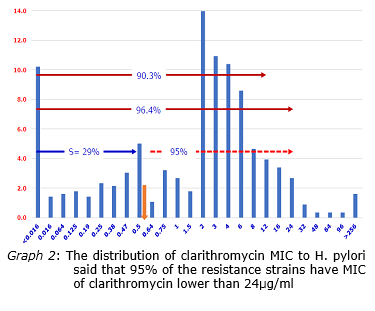
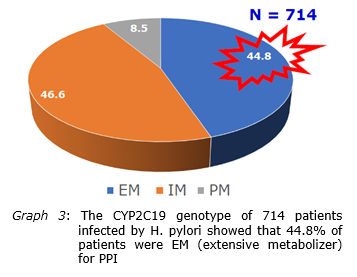
In 2016 there were 1511 cases of H. pylori were isolated in which 894 were from children and 617 were from adults. In terms of antimicrobial resistance, the results showed 71% of the isolates resistant to clarithromycin, 45% metronidazole resistance, 32% levofloxacin resistance, 16% tetracycline resistance, and 17% amoxicillin resistance. The results also showed that MIC90 of clarithromycin was 12μg/ml and thus, in terms of pharmacokinetics and pharmacokinetics of the gastric clarithromycin (rapid dissipation in the first 30 minutes in the stomach), H. pylori was still sensitive to this kind clarithromycin. Analysis of the distribution the clarithromycin MIC, 96.4% H. pylori had the MIC of 24μg/ml or less and clarithromycin was able to reach this concentration. The H. pylori multi-drug resistance analysis revealed 1% the isolates resistant to all antibiotics, 3% resistant to four antibiotics, 18% resistant to three antibiotics, 41% resistant to two antibiotics. 26% were resistant to 1 antibiotic, and 11% were completely antibiotic-susceptible. Analysis of the virulent genotype of the H. pylori isolates resulted 77% of H. pylori isolates from adults or children carrying the cagA gene. In H. pylori existing cagA gene, vacA gene in isolates from adults and children was 60.4% and 69.4% s1m1, 39.3% and 30% s1m2, 0.3% and 0.1% s2m1, 0% and 0.4% s2m2. Analysis of the CYP2C19 genotype, results showed that 44.8% were extentive metabolism, 46.6% were moderate metabolism, 8.5% were poor metabolism.
CONCLUSION
This is a comprehensive study in Vietnam on microbiology and molecular biology on a large number of H. pylori isolated from gastro-duodenal ulcer biopsies. The most notable result of this study is that H. pylori in Vietnam has a relatively high resistance to antibiotics, especially clarithromycin and metronidazole. However, the level of clarithromycin resistance is not high because the clarithromycin MICs to H. pylori are still lower than the clarithromycin concentration achieved in the stomach if clarithromycin is used for the treatment is the gastric clarithromycin, not pulmonary clarithromycin.
Other research
- Overdiagnosis: When the Boundaries of Disease Become Blurred ( 08:22 - 08/07/2025 )
- Continuous Medical Knowledge Tied to Better Clinical Outcomes in Internal Medicine ( 10:56 - 19/06/2025 )
- Rapid and sensitive diagnostic procedure for multiple detection of pandemic Coronaviridae family members SARS-CoV-2, SARS-CoV, MERS-CoV and HCoV: a translational research and cooperation between the Phan Chau Trinh University in Vietnam and Universit ( 15:42 - 29/06/2020 )
- PCTU studies at ISSAR 2019 international conference - Gyeongju, Korea ( 16:17 - 04/11/2019 )
- Melioidosis in Vietnam: Recently Improved Recognition but still an Uncertain Disease Burden after Almost a Century of Reporting ( 13:29 - 27/04/2018 )
- Microbiological agents causing community pneumonia require hospitalization ( 11:20 - 03/02/2018 )
- The in-vitro activity of sitafloxacin against the most common bacterial pathogens isolated from the clinical samples ( 11:10 - 03/02/2018 )
- Evaluate the role of the microbiological molecular biology tests in the early detection of pathogens causing lower respiratory infections ( 10:52 - 03/02/2018 )
- Production and evaluation of the kit using magnetic silica coated nano-iron beads to extract the nucleic acid from different samples ( 10:16 - 03/02/2018 )
- The resistance of the gram-negative rods isolated from clinical cases in Vietnam to doripenem ( 08:46 - 03/02/2018 )